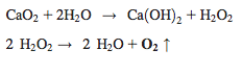|
Chandra et al. developed a wound dressing incorporating particulate oxygen generators (POG) to deliver oxygen to the wound in situ [1]. The dressing consists of 4 layers: gelatin (layer 1), sodium percarbonate (SPO) and calcium peroxide (CPO) in a polycaprolactone (PCL) and polyvinyl alcohol (PVA) matrix (layer 2), silicone (layer 3), and polyvinylidene chloride (PVDC) (layer 4) [1]. The purpose of the gelatin layer is to absorb the wound exudate [1]. Once the exudate passes through the first layer, the SPO and CPO undergo decomposition to release oxygen (figure 3.1)[1]. Hydrogen peroxide is an intermediate in this reaction and can be forced to decompose into oxygen in an alkaline environment [1]. To further assure this decomposition occurs, manganese chloride was incorporated into the gelatin layer to catalyze decomposition [1]. The silicone layer provides mechanical strength and flexibility to the dressing [1]. The PVDC layer allows for low gas and vapor permeability ensuring the oxygen diffuses in the direction of the wound [1]. The distribution of SPO and CPO in layer two was confirmed to be uniform by scanning electron microscope analysis [1]. Oxygen release studies were performed by placing the second layer in either a water rich environment or a water depleted environment [1]. In the water rich environment, 70% of the oxygen was released during the first 24 hours [1]. After 24 hours, the amount of oxygen released increased and remained constant for the next 48 hours [1]. In the low water environment, oxygen release increased for the first 24 hours and remained constant for the following 48 [1].
A porcine full-thickness surgical wound model was implemented in this study [1]. Pig skin tissue was surgically removed to create a wound [1]. By week 1, the wound was filled with collagen and fibronectin extracellular matrix [1]. By week 2, the healing process had begun and the wound began to close [1]. In the case of the POG dressing, the tissue was better vascularized and the wound edges were closing in a more uniform matter when compared to the controls [1]. To quantify this data, relative wound closure and reepithelialization were calculated from the wound images [1]. From this analysis, the researchers determined that the wounds closed faster and that reepithelialization occurred faster for the POG group [1]. Increasing the rate of wound closure and reepithelialization results in faster tissue healing and lower infection rates [1]. The researchers then tested the quality of the regenerated tissue by performing Hematoxylin-Eosin (H&E) and Masson-Trichrome staining [1]. In the POG treated groups, a hyperproliferative epidermis at the wound edges was observed in week 4 [1]. By the time it was week 6, the layer had compressed to form the epidermal papillae [1]. At week 8, it was even thinner and the structure resembles that seen in normal pig skin [1]. The control was not hyperproliferative by week 4 and at week 8 the epidermal papillae could not be seen meaning the epidermal layer had not yet matured [3.1]. Collagen deposition is important for providing the matrix for tissue remodeling and angiogenesis [1]. The researchers measured the amount of collagen in the upper dermis (UD), middle dermis (MD), and lower dermis (LD) [1]. They concluded that the amount of collagen in each layer changes over an 8-week period [1]. To determine the amount of angiogenesis, they examined the formation of new blood vessels using staining for the von Willebrand Factor (vWF) and the CD31 antibody [1]. The POG dressing showed a higher density of new blood vessels compared to the control in all three layers of dermis by both stainings [1]. The POG dressing had more small diameter vessels in the UD and LD than the control in week 2 [1]. By week 8, the number of blood vessels had decreased and the LD had a substantial amount of large diameter vessels [1]. This was not observed in the control dressing [1]. The advantages of using this technology in our design include: faster wound closure, faster reepithelialization, and structure resembling that of normal big skin. The limitations of using this approach to aid diabetic foot ulcers is that it has not yet been tested on a diabetic porcine model. It is hard to tell whether the oxygen delivery will be enough to move the healing process out of the inflammation phase that diabetics often get caught in. Zhou et al. prepared carboxyethyl chitosan / poly (vinyl alcohol) (CECS/PVA) nanofibers via electrospinning as a potential material for wound dressings [2]. Mouse fibroblasts (L929) were used to evaluate this material in vitro for skin regeneration [2]. Cytotoxicity was measured indirectly and indicated that the material was non-toxic to the fibroblasts [2]. Cell culturing indicated that the material promoted L929 cell attachment and proliferation [2]. Ignatova et al. studied photo-crosslinked electrospun mats containing quaternary chitosan (QCS) and PVA [2]. The mat inhibited gram-positive and gram-negative bacteria suggesting promise for wound-dressing applications [2]. Yang et al. proposed hydrogels made of PVA, water-soluble chitosan, and glycerol fabricated by irradiation followed by freeze thawing as a potential wound dressing [2]. An MTT assay indicated that the hydrogels were nontoxic towards L929 [2]. They indicated that the material accelerated the healing process in a rat model [2]. Other findings showed that the wounds treated with this material healed by the 11th day and showed mature epidermal architecture [2]. Thus, this material shows promise as a wound dressing material [2]. Sung et al. introduced minocycline-loaded wound dressings: cross-linked hydrogel films prepared with PVA and chitosan via freeze-drying method [2]. Chitosan was shown to decrease the gel fraction, maximum strength, and thermal stability of the PVA hydrogel [2]. On the other hand, it increased the swelling ability, water vapor transmission rate, elasticity, and porosity [2]. References [1] Chandra, P. K. et al. “Peroxide-based oxygen generating topical wound dressings for enhancing healing of dermal wounds,” Wound Healing Society, vol. 23, pp. 830-841, 2015. [2] R. Jayakumar, M. Prabaharan, P. S. Kumar, S. Nair, and H. Tamura, "Biomaterials based on chitin and chitosan in wound dressing applications," Biotechnology advances, vol. 29, pp. 322-337, 2011.
0 Comments
|
|